|
Problem description states that, we need to apply Le Chatelier's principle, for aqueous Cobalt chloride reaction. If you have any questions, don't hesitate to ask. Point to Stockroom : Stockroom Explorer has required solutions and Problem Description. Older PDF version is included in the G-Drive directory.įeel free to edit as needed and share with your students in whatever form you wish. Reformatted for 2022 as an editable Google Doc! The newest version changes the order of the EQ stresses and adds a common ion section with additions of AgNO3 and NaOH to precipitate the cobalt ion. This is my 2022 introduction of the *old* version of the lab. Materials needed: cobalt chloride solid, E-flasks, HCl (4-8 M), transfer pipettes or droppers, heat source, ice. Learning Target Log Update Update your LT Log for LT’s 8.7-8.In this lab students will create a complex ion equilibrium of cobalt chloride and then stress the equilibrium observing changes as predicted by LeChatelier's principle. 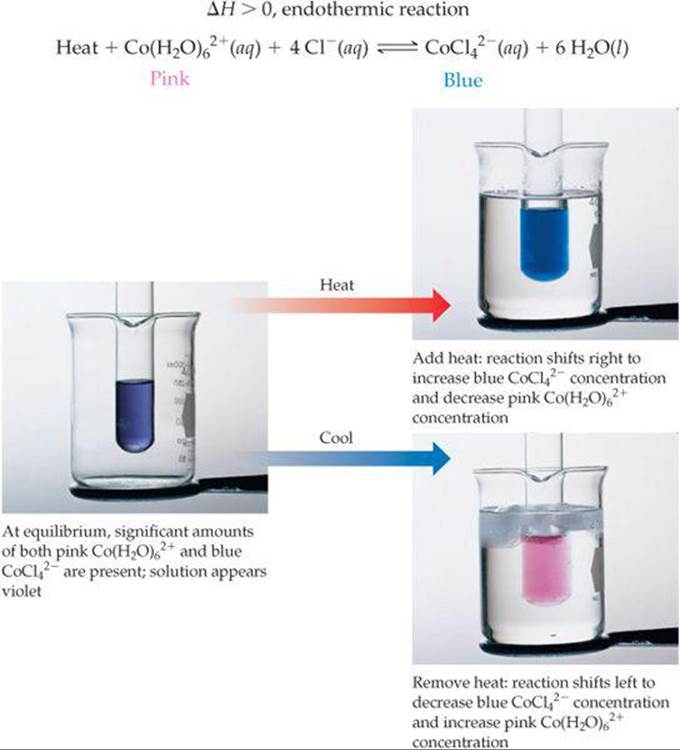
Therefore, based on the color of the reaction mixture we can conjecture if Keq is greater than or less than 1.” Predict how the addition of sodium chloride would affect the equilibrium. add heat to left side, heat is +50 kJ/mol. Keq 1 the equilibrium lies to the right, and if Keq Keq > 1: Products favored at equilibrium. 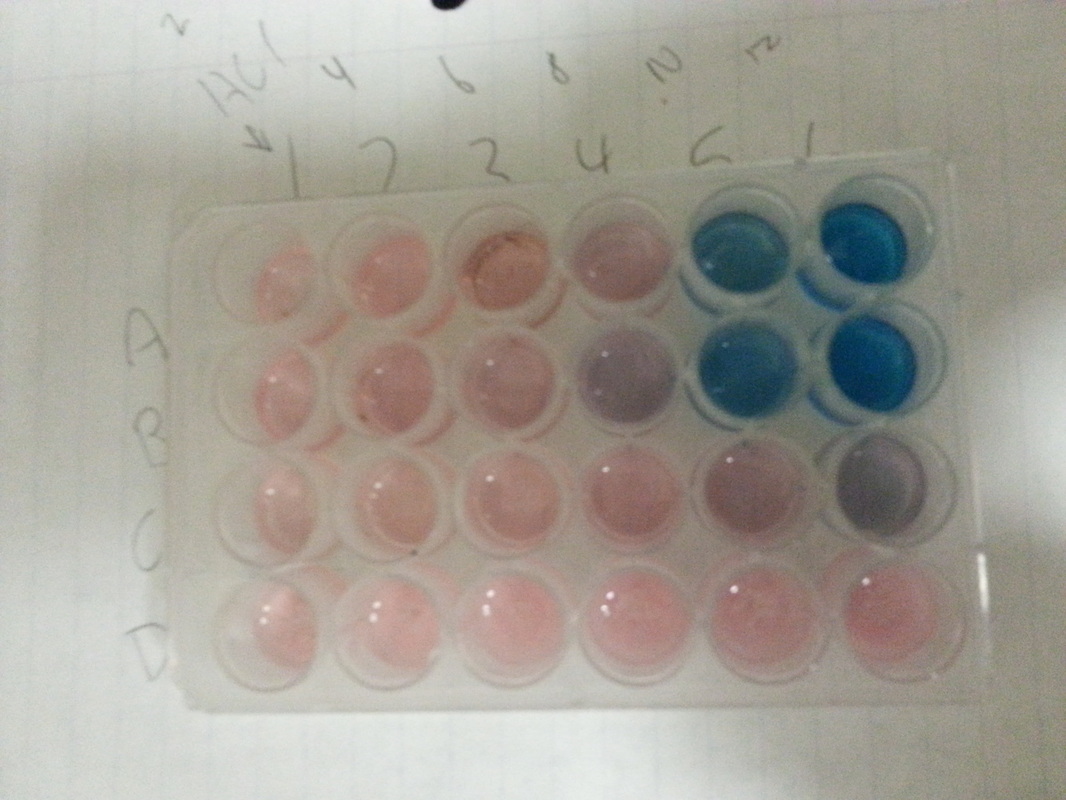
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
